Non-Invasive Glucose Sensing: Why the Hardest Part Is Integration
A first-principles architectural thesis: non-invasive glucose monitoring has remained open for twenty-five years not because any single domain is unsolved, but because the moat is integration across optics, tissue physiology, silicon photonics, thermal control, and inference.
This is a first-principles architectural thesis. Numerical claims trace to cited literature; architectural claims are the author’s reasoning, offered for critique rather than as established fact. The figures labeled “conceptual schematic” depict proposed design choices, not measured layouts.
Twenty-five years after the FDA approved Cygnus’s GlucoWatch, a truly non-invasive glucose monitor has still not reached the market.
That fact is worth sitting with.
The failure is not for lack of effort. Serious teams have tried. Cygnus used reverse iontophoresis to pull glucose-bearing interstitial fluid across the skin. It reached the market, then disappeared. ArKal pursued hollow microneedles and demonstrated clinical feasibility, but tissue response limited wear time. Google and Novartis explored glucose sensing through tears in a contact lens and eventually stopped the program. Today’s commercial CGMs, including Dexcom G7 and Abbott FreeStyle Libre 3, are remarkable products, but they are still invasive. Their electrodes sit under the skin. Biolinq’s recently cleared microneedle patch is shallower and elegant, but it remains minimally invasive.
The goal that remains open is harder. Measure glucose continuously, through intact skin, without drawing blood, inserting a filament, or penetrating tissue.
The question is not whether glucose has an optical signature. It does. The question is whether that signature can be separated, reliably and repeatedly, from the much larger and more variable biological background of living skin.
That is the real problem.
The problem is not detection. It is separation.
Every sensing problem is a signal-to-noise problem.
For non-invasive glucose, the signal is small but not vanishingly small. In the 1500 to 1700 nm near-infrared window, glucose has absorption features associated with C–H first-overtone bands. At physiological concentration around 5 mM, the glucose absorbance over a roughly 1 mm dermal path is on the order of to [1]. That is detectable with modern photodiodes and modest averaging.
So the hard part is not detecting a weak signal. The hard part is that glucose sits inside a biological matrix whose optical contribution is much larger.
Water is present at roughly 55 M and absorbs strongly in the same band. Lipids, hemoglobin, melanin, collagen, albumin, and other constituents add their own spectral signatures. More importantly, they vary. Hydration changes over the day. Perfusion changes with meals, exercise, temperature, stress, and circadian rhythm. Skin thickness, melanin, fat, vascularity, and scattering vary across people. Contact pressure from the device changes the optical path. Motion changes the tissue volume being sampled.
The glucose signal is a small coherent chemical signature sitting on a large, moving, person-specific biological background.
That distinction matters. If this were a problem of photon sensitivity, better detectors would solve it. The obstacle is not the absolute size of the glucose signal. It is the variability of everything else.
A 10% change in local hydration can shift the water contribution by more than the entire glucose signal being measured.
That is the arithmetic that has defeated the field.
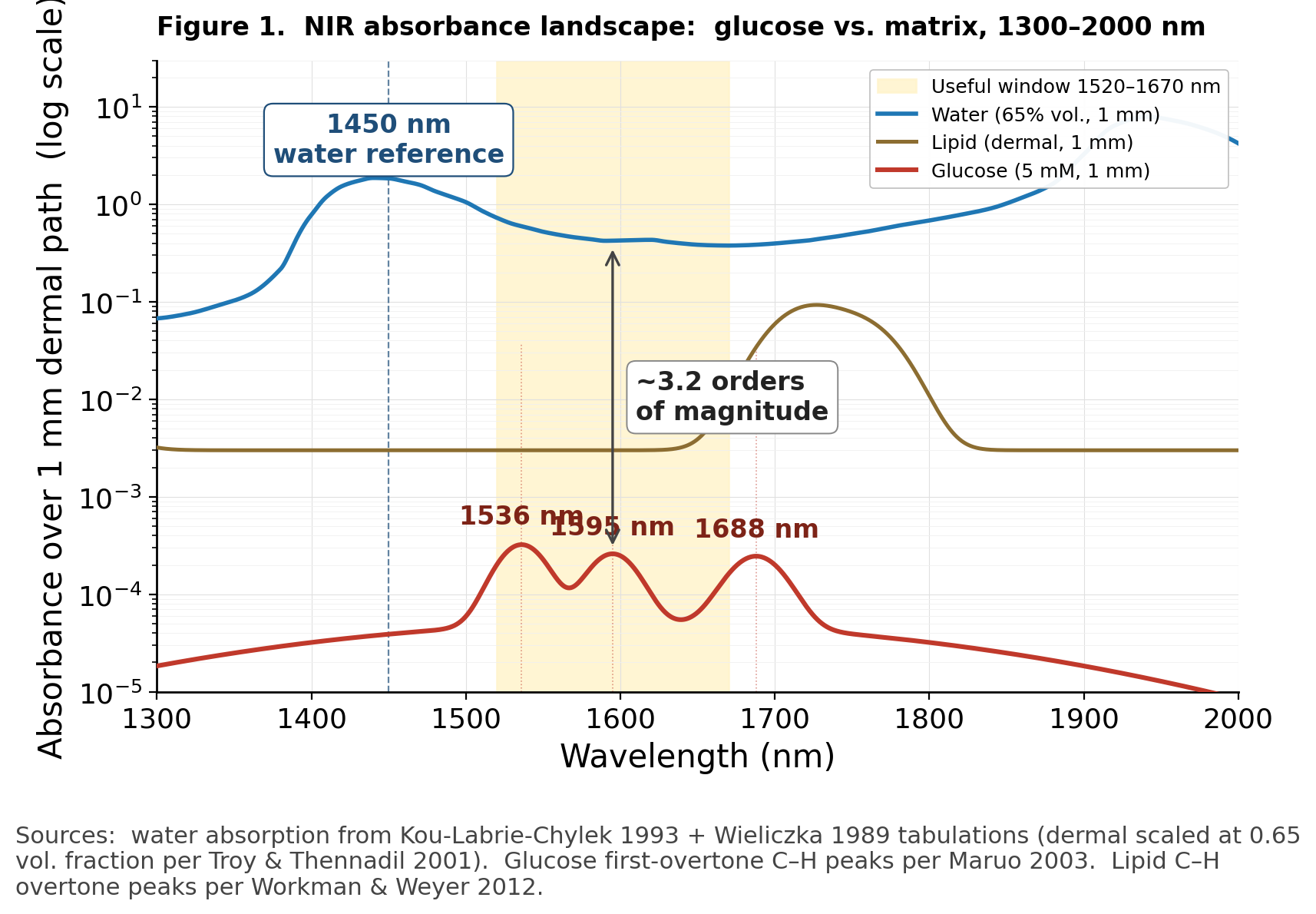
Figure 1. The signal-to-background problem at first sight: glucose absorbance is three to four orders of magnitude below water across the working NIR band. Overlaid log-scale absorbance over a 1 mm dermal path between 1300 and 2000 nm for water (blue), dermal lipid (olive), and physiological glucose at 5 mM (red). Water curve scaled to 65% volume fraction in dermis (Troy & Thennadil 2001) using the absorption-coefficient tabulations of Kou, Labrie & Chylek 1993 and Wieliczka, Weng & Querry 1989. Glucose first-overtone C–H peaks at 1536, 1595, and 1688 nm modeled as Gaussians (35–40 nm FWHM) with molar absorptivities from Maruo et al. 2003. Lipid peaks at 1210 and 1720 nm scaled to dermal volume fraction per Workman & Weyer 2012. Soft yellow band marks the useful spectroscopic window (1520–1670 nm) where glucose signal strength and dermal photon penetration overlap. Dashed line at 1450 nm marks the water-reference channel that anchors the architecture’s calibration. Annotated arrow at 1595 nm marks the ~3.2 orders of magnitude that separates the matrix bulk from the analyte signal. That gap is what the architecture’s selectivity, reference anchoring, and physics-informed inversion stack have to navigate together.
This problem class is not unique to glucose. Environmental gas spectroscopy faces the same dominant interferent in atmospheric water vapor, alongside CO₂ and methane, and most of the engineering effort there goes to selectivity at the source, deterministic correction for the matrix, and constrained inference rather than raw detection sensitivity. The lessons transfer. What makes the wrist version harder is that the matrix is alive, viscoelastic, vascular, and circadian, and the geometry is fixed at the dermal thickness with no path-length lever.
The three levers
Once the problem is framed as signal-to-noise, the design space becomes clearer. A successful architecture must use three levers together.
Amplify the glucose contribution relative to the background. In a benchtop cuvette one could increase path length. On the wrist the optical path is constrained by tissue geometry. The practical amplification lever is selectivity. Use narrow-linewidth sources at wavelengths where glucose absorbs and where the interfering matrix is less damaging.
Reduce matrix variability before it reaches the inference engine. That means thoughtful source-detector geometry, multi-distance measurements, ratio channels, reference wavelengths, explicit temperature measurement, hydration estimation, perfusion sensing, and motion or pressure gating.
Extract the residual glucose signal using constrained inference. This is where modern machine learning can help, but only after the physical measurement has been made as specific and stable as possible. AI cannot rescue an instrument that has not captured glucose-specific information. It can only help separate glucose from residual confounders when the measurement already contains enough physics.
The trap is to believe any one lever is enough. A clever wavelength is not enough. A beautiful photonic chip is not enough. A deep neural network is not enough. A better contact interface is not enough. A new packaging concept is not enough. The problem rewards integration.
Why wavelength selectivity matters
For optical glucose sensing, the source is not an implementation detail. It is the beginning of the measurement.
Glucose has useful first-overtone absorption features in the 1500 to 1700 nm region, with peaks at approximately 1536, 1595, and 1688 nm [1]. The window is attractive because the glucose signal is stronger than in the higher-overtone region while penetration is still sufficient to reach the dermal compartment. Move shorter and glucose absorption becomes too weak. Move longer and water absorption grows so strongly that the photon budget collapses. The usable band is narrow, roughly 1520 to 1670 nm, with a dedicated water reference channel near 1450 nm where the water peak is bright and well-characterized.
Within that band, broad sources are problematic. The cleanest first-overtone glucose features sit at roughly 40 to 80 nm FWHM. A source much wider than that averages over glucose, water, lipid, and other tissue contributions; the selectivity is washed out before the detector ever sees the light.
A narrow-linewidth source is different. A laser source like a VCSEL uses an optical cavity to define wavelength and linewidth. Spontaneous emission in a direct-bandgap III–V at the ~0.8 eV bandgap that corresponds to 1.55 µm emission broadens to 50 to 100+ nm at room temperature from thermal-population effects across the gain spectrum. The optical cavity, defined by the DBR mirrors above and below the active region, narrows that to sub-nm to a few nm. A laser is a spectrally clean source by construction; an LED is not.
The VCSEL platform is also industrialized at NIR consumer scale. The 940 nm VCSEL platform ships in billions of units in iPhone Face ID, indirect time-of-flight depth sensors, and automotive LiDAR. The 850 nm platform underlies multimode datacom for two decades. The 1310 to 1550 nm platform supports telecom CWDM. The 1500 to 1700 nm regime needed for glucose is harder. It requires InP-based active regions because GaAs cannot reach the bandgaps. Two routes exist. Monolithic InP MQW VCSELs grown with InP-lattice-matched DBRs have low refractive-index contrast, which forces high mirror layer counts and limits yield. Wafer-bonded VCSELs use an InP gain region bonded to high-contrast GaAs-based mirrors, currently shipping at telecom-grade volumes in CWDM transceivers. Consumer-volume reliability at 1500 to 1700 nm is established at telecom-grade specifications but not yet at iPhone-camera reliability. A glucose program operating in this regime is buying telecom-grade VCSEL platform maturity, not consumer-grade. That gap is real and budgetable.
LED-class sources at these wavelengths exist as discrete devices but are not viable here. Auger recombination at narrow bandgaps below ~0.85 eV degrades wall-plug efficiency, the intrinsic spontaneous-emission FWHM at these bandgaps is wide, and there is no display-driven volume economics at 1500 nm to fund the chip-scale industrialization that visible micro-LEDs received. The III-nitride platform that transformed visible micro-LEDs caps at ~650 nm and does not reach this window at all.
The likely architecture is not one magic tunable laser sweeping the entire band. Thermal tuning a single VCSEL across 100 nm would require approximately 1300°C of swing, which is impossible at any worn-device thermal budget. The plausible architecture is a multi-element fixed-wavelength array. Several VCSELs at carefully chosen glucose and reference wavelengths, each thermally controlled over a small range.
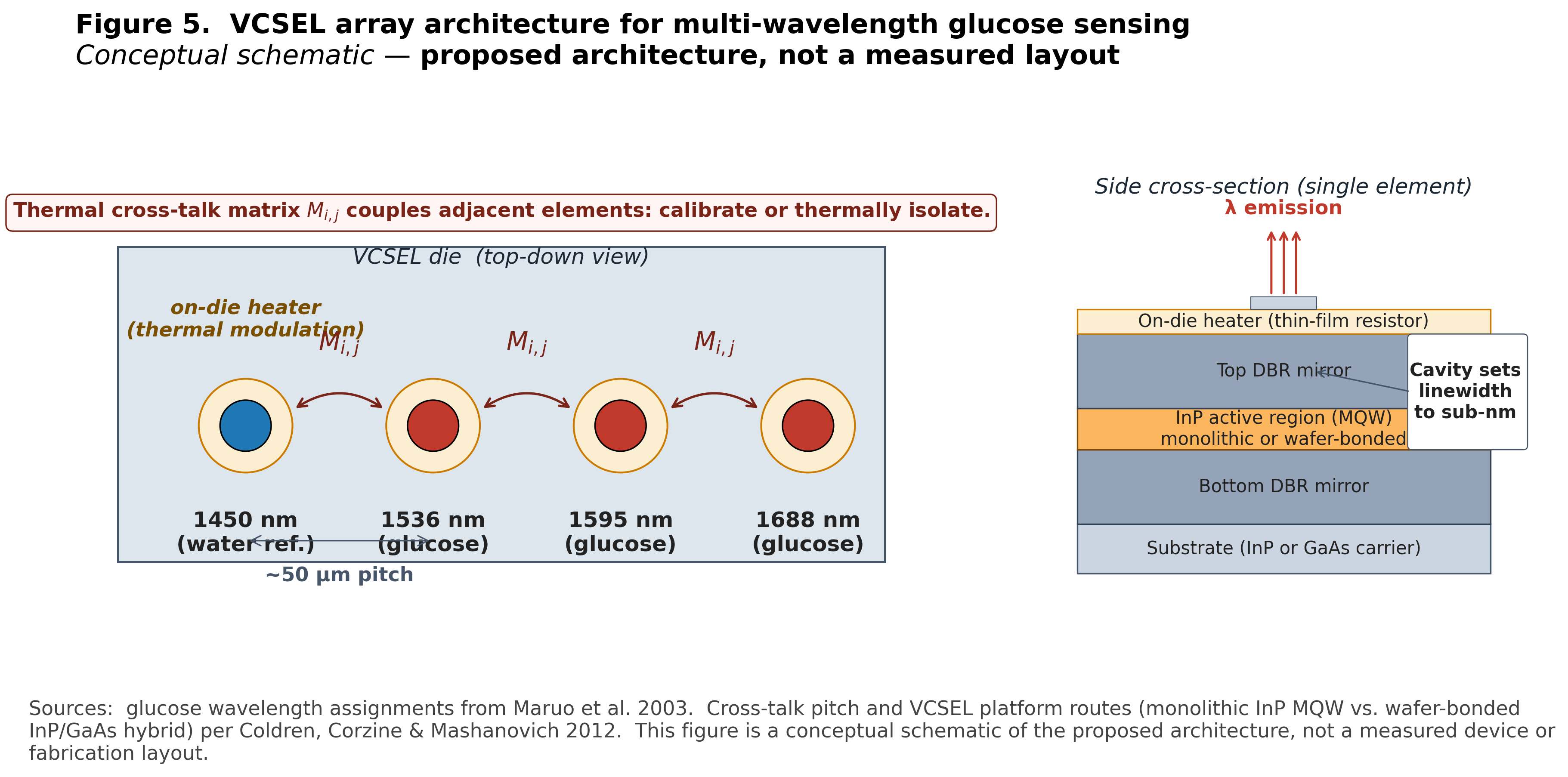
Figure 5. A four-element fixed-wavelength VCSEL array with on-die thermal modulation and a calibrated cross-talk matrix is the architecturally coherent source for first-overtone glucose spectroscopy. Top-down schematic (left) shows four VCSEL elements at 1450 nm (water reference channel), 1536 nm, 1595 nm, and 1688 nm (the three glucose first-overtone C–H peaks of Maruo et al. 2003), each surrounded by an on-die thin-film resistor heater for ~1–5°C of per-element thermal modulation around the design-center wavelength. Curved double-headed arrows mark the inter-element thermal cross-talk matrix that adjacent ~50 µm-pitch heaters impose on each other; is bounded (~10–30% of self-coupling at this pitch) and is either eliminated by per-element trench / backside-thinning isolation or absorbed via active calibration with on-board reference temperatures. Side cross-section (right) shows the layer stack: substrate (InP or GaAs carrier), bottom DBR mirror, InP active region with multi-quantum-well gain (or a wafer-bonded InP active region on a GaAs DBR for higher-contrast mirrors per the platform routes in Coldren, Corzine & Mashanovich 2012), top DBR mirror, integrated heater, with the optical aperture emitting upward through the top DBR. The cavity formed between the two DBRs is what reduces emission linewidth from the 50–100+ nm FWHM of spontaneous-emission-class sources to the sub-nm linewidth that first-overtone glucose spectroscopy requires. Conceptual schematic of the proposed architecture, not a fabrication layout or measured device.
That choice creates a systems problem.
A VCSEL’s wavelength shifts with temperature at to nm/°C [2], driven by the cavity refractive-index temperature coefficient and DBR thermal expansion. Useful operating modes need ~0.1 nm modulation around peak (achievable with ~1.5°C of thermal swing) and ~1 nm hops between peak and flank for ratio measurement (~12 to 15°C). Each element therefore needs 1 to 5°C of active control, achievable with on-die heaters dissipating milliwatts. Heat from one heater diffuses through the substrate and shifts neighboring elements. Cross-talk at ~50 µm pitch is on the order of 10 to 30% of self-coupling and forces either thermal isolation through trenches or active calibration of a cross-talk matrix that maps heater currents to per-element wavelength shifts.
Skin temperature also shifts the absorption peaks of water and glucose, in known but coupled ways. Water O–H overtones shift at ~0.2 to 0.3 nm/°C, glucose C–H overtones at ~0.05 to 0.1 nm/°C [3]. Over a 7°C wrist swing the water peak moves ~1.75 nm and the glucose peak ~0.53 nm, both at or beyond the precision the inversion requires. The measurement now depends on at least three temperature states. VCSEL die temperature, skin temperature, and ambient temperature, all measured and propagated through the calibration pipeline.
None of this is fatal. But it must be designed into the architecture from the beginning.
This is the pattern of the entire problem. A promising idea in one domain becomes usable only when its implications are carried through the adjacent domains.
Skin is not a passive sample holder
The wrist is not a cuvette.
Skin is layered, scattering, vascular, pigmented, viscoelastic, hydrated, and alive. The stratum corneum is dry and highly scattering. The viable epidermis is hydrated and avascular. The dermis contains the capillary bed and the collagen-rich matrix. Subcutaneous fat changes the optical boundary conditions. Melanin varies across skin types. Blood volume changes with physiology.
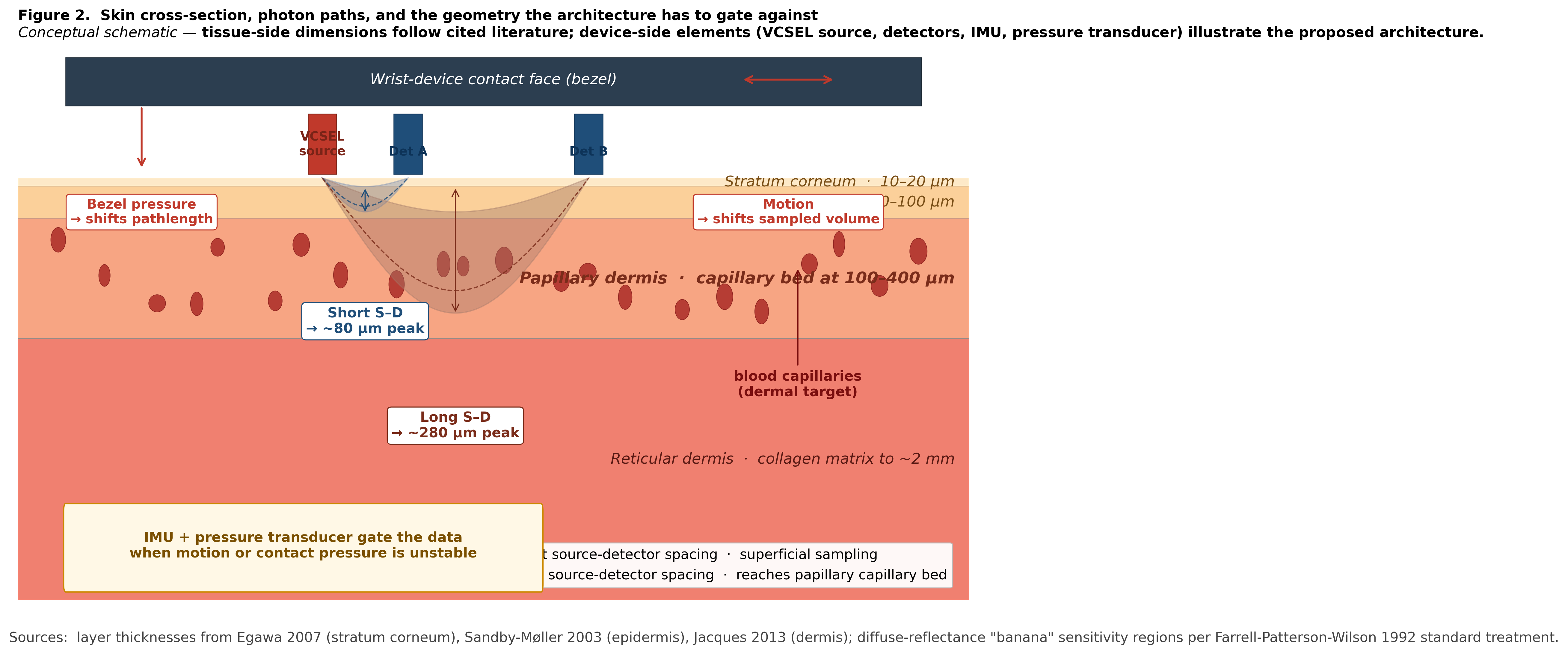
Figure 2. The wrist is a layered, vascular, viscoelastic optical environment whose geometry shifts with motion and pressure. Cross-section through skin showing the stratum corneum (10–20 µm; Egawa et al. 2007), viable epidermis (30–100 µm; Sandby-Møller et al. 2003), papillary dermis with the capillary bed at 100–400 µm depth, and reticular dermis (Jacques 2013). Capillary loops shown in the papillary dermis are the dermal targets the architecture interrogates. Wrist-device contact face (bezel) sits above the surface; a VCSEL source and two detectors at different source-detector spacings illustrate how short S–D paths sample superficial layers (~80 µm peak depth) while longer S–D paths reach the papillary capillary bed (~280 µm peak), per the diffuse-reflectance “banana” geometry of Farrell, Patterson & Wilson 1992. Red arrows indicate the two principal noise vectors the optical channel cannot correct internally: lateral motion shifts the sampled tissue volume; bezel pressure compresses tissue and shifts the effective optical pathlength. The IMU + pressure transducer subsystem (lower-left callout) gates the optical samples when these vectors exceed thresholds, accepting reduced duty cycle as the cost of avoiding contaminated training data. Conceptual schematic. Tissue-side dimensions follow cited literature; device-side elements illustrate the proposed architecture and are not a fabrication layout.
The tissue also responds mechanically. Contact pressure changes the optical path length. Compression alters scattering and local blood volume. Hydration changes both the mechanical response of skin and the water absorption baseline. Temperature shifts both tissue physiology and spectral peak positions.
These effects are coupled. Treating mechanics, optics, physiology, and temperature as independent error terms misses the structure of the problem.
This is why motion and contact pressure matter so much. If the wrist device shifts, or the bezel pressure changes, the photons no longer sample the same tissue volume. The resulting optical signal may be real, but it belongs to a different forward problem. A machine-learning model trained as if the geometry were constant will happily absorb those artifacts as apparent glucose information.
That is how plausible-looking models fail in the field.
A serious architecture must therefore include state awareness. Inertial sensing, contact pressure, temperature, perfusion, and the optical channels themselves are not optional accessories. They determine when the optical measurement is trustworthy and when it should be gated out. Gating reduces duty cycle. That is the cost. But using bad data is worse. In a medical device, a confident wrong answer is more dangerous than no answer.
Where machine learning helps, and where it does not
Machine learning is powerful in this problem and often discussed too casually.
Classical chemometrics, such as partial least squares regression, can find correlations between spectral features and glucose. The danger is that it can also find correlations between confounders and glucose. If post-meal glucose rise is correlated with skin temperature, perfusion, or hydration in the training set, the model learns those variables as glucose proxies. It performs well in-sample and fails when deployed to a different person, environment, or routine. The FDA has cited this failure mode specifically [4] when evaluating non-invasive submissions. Deep learning does not automatically solve this. It can learn spurious proxies more efficiently.
The right role for AI in this problem is narrower and more valuable. Constrain the inference using physics.
Take the largest single coherent confound, which is thermal-spectral coupling. Skin temperature shifts the spectral behavior of water and glucose in non-arbitrary ways.
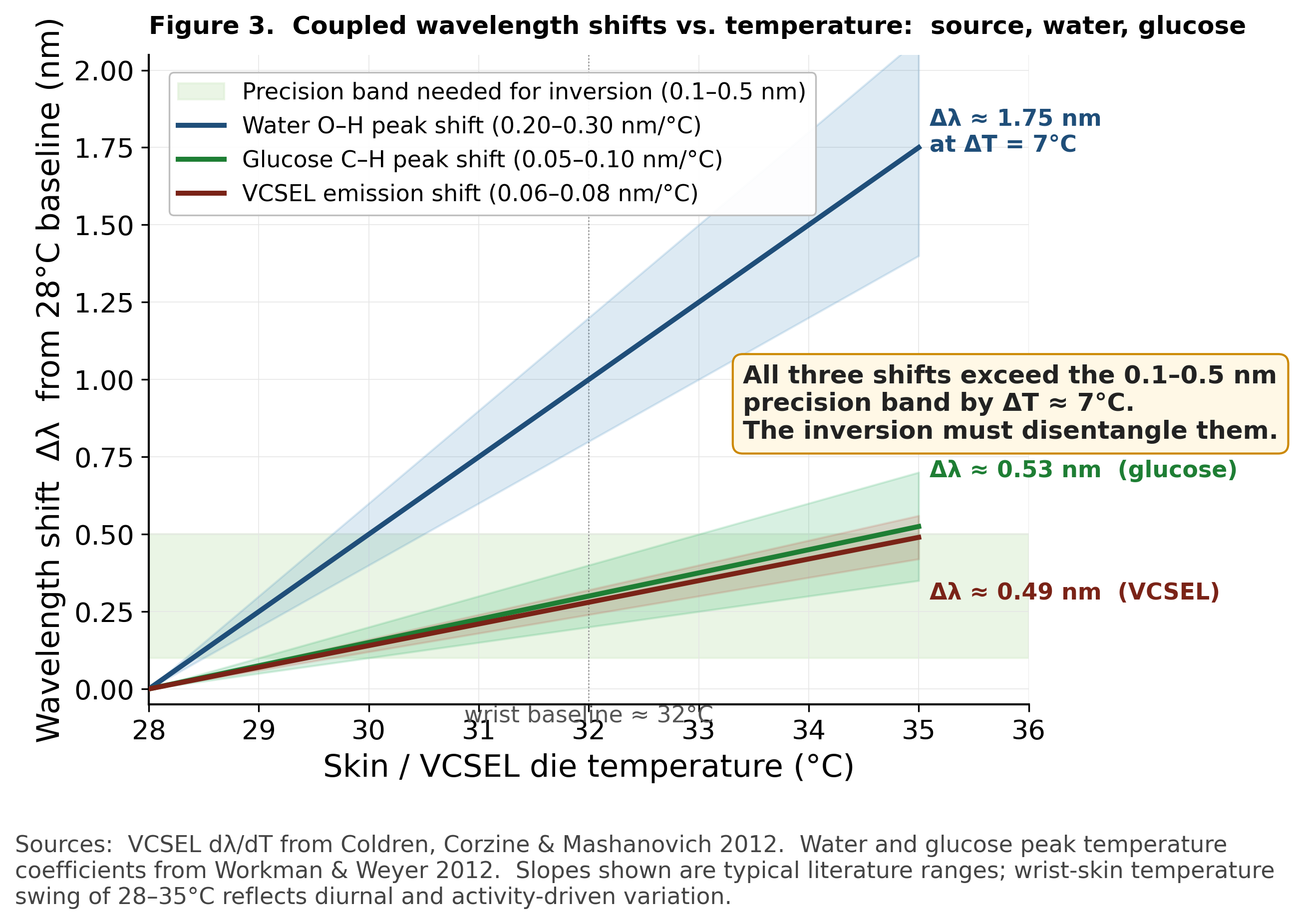
Figure 3. Three coupled wavelength shifts driven by temperature, all exceeding the inversion-precision band by ΔT ≈ 7°C. Wavelength deviation Δλ from the 28°C baseline as a function of skin or VCSEL die temperature, computed for VCSEL emission (red, = 0.06–0.08 nm/°C; Coldren, Corzine & Mashanovich 2012), water O–H first-overtone peak position (blue, 0.20–0.30 nm/°C; Workman & Weyer 2012), and glucose C–H first-overtone peak position (green, 0.05–0.10 nm/°C; Workman & Weyer 2012). Shaded bands around each line show the literature range; center curves use the band midpoint. Soft green horizontal band at Δλ = 0.1–0.5 nm marks the precision the quantitative inversion can tolerate (~10% of the glucose peak FWHM). At a typical 7°C wrist-skin range from baseline (28°C → 35°C), the water peak shifts ~1.75 nm, the glucose peak ~0.53 nm, and the source emission ~0.49 nm. All three are above the tolerance band. All three are coupled. All three move at different rates. The Thermal-Spectral Constraint Engine encoded below addresses exactly this drift: it forces the inference network to respect the deterministic temperature dependence rather than absorb the coupled drift as opaque covariance. The “wrist baseline ≈ 32°C” dotted vertical marks the typical steady-state worn-device skin temperature.
A physics-informed model encodes that thermal-spectral coupling as a loss-function constraint rather than learning it. The architecture pairs each glucose-linked wavelength nm with a 1450 nm water reference and writes a thermal-spectral constraint into the training:
The constraint forces the model’s predicted spectrum-temperature gradient at glucose-linked wavelengths to track the temperature dependence of the water peak, scaled by the molar extinction ratio between them. The literature extinction values [1] enter this term as priors on the ratio, not as hard physical laws. Modified Beer-Lambert in scattering tissue, with a wavelength-dependent differential pathlength factor [5], means the in-vivo ratio differs from the in-vitro literature, and the network must be free to update within bounded ranges as it sees calibration data. The constraint shrinks the hypothesis space from “any mapping from spectra to glucose” to “mappings whose thermal behavior is consistent with the known temperature physics of water.” The spurious-proxy failure mode is not eliminated, but the largest contributor to it is removed.
That is the promise of physics-informed ML here. It does not replace source selectivity, optical design, calibration, or physiology. It sits on top of them.
In that role, AI is leverage. Used as a substitute for instrumentation, it is a liability.
What the history teaches
The history of glucose sensing makes more sense through the signal-to-noise lens.
Cygnus tried to bring glucose to the skin surface using reverse iontophoresis. It solved non-invasive access partially but created skin irritation and reliability challenges at useful currents. ArKal used hollow microneedles to access interstitial fluid more directly. It improved access but ran into tissue response around the needles. Dexcom and Abbott accepted invasiveness and engineered the tissue-electrochemistry interface through enzyme chemistry, membranes, calibration, algorithms, and manufacturing discipline. They shipped because their architecture made the signal accessible and managed the biological interface well enough over wear time. Biolinq moved shallower, into the epidermis, with redundant microneedle electrodes at each tip. The trade was less depth, less invasiveness, and redundancy at the sensing interface, and that combination unlocked a week of wear. The contact-lens approach pursued tears as a non-invasive fluid. The challenge was not detecting glucose in tears. It was making tear glucose a reliable proxy for blood glucose across time, physiology, and environment.
Optical wrist sensing makes a different bet. It avoids penetration entirely but inherits the full complexity of diffuse reflectance through living skin. That is the most ambitious version of the problem.
Every architecture spends its signal-to-noise budget somewhere. The products that ship are the ones whose spending matches what the physics can actually deliver.
From proof of concept to product
Non-invasive glucose has had proof-of-concept demonstrations for decades. That does not mean the product is close.
Proof of concept shows that glucose-dependent signal can be measured under controlled conditions. Clinical validation shows that the measurement works across a meaningful population under less controlled conditions. Commercial deployment requires the system to work across the full real-world envelope. Skin types, activity levels, meals, exercise, temperature, sweat, pressure, motion, manufacturing variation, calibration drift, software updates, and wear-life changes.
The last step is where many technologies fail. It is also where most of the real engineering lives.
A non-invasive architecture that integrates source selectivity, geometry and reference anchoring, thermal-spectral decoupling, and motion / contact-pressure state-gating earns honest accuracy bands at each phase. In controlled sedentary cohorts (subjects at rest, stable thermal state, gated wear) the architecture is dimensioned to deliver MARD in the 9 to 10% range, the FDA iCGM Special Controls threshold [6]. This is the range the published non-invasive literature has occasionally landed under controlled conditions. The architectural claim is that those occasional landings become reproducible when all the levers are integrated, not when one or two carry the system. A representative real-world MARD is anticipated at 15 to 20%, accounting for the residual noise of the sensor-skin interface during active use that no architecture eliminates entirely. The gap is not a defect. It is the cost of operating on a wrist instead of a benchtop. Closing the gap to the iCGM bar across the deployment envelope is what Phase 3 engineering looks like for this class of device.
For reference, Dexcom G7 reports MARD ~8% [7] and FreeStyle Libre 3 similar [8]. These are outputs of more than a decade of serious Phase-3 iteration on an invasive architecture.
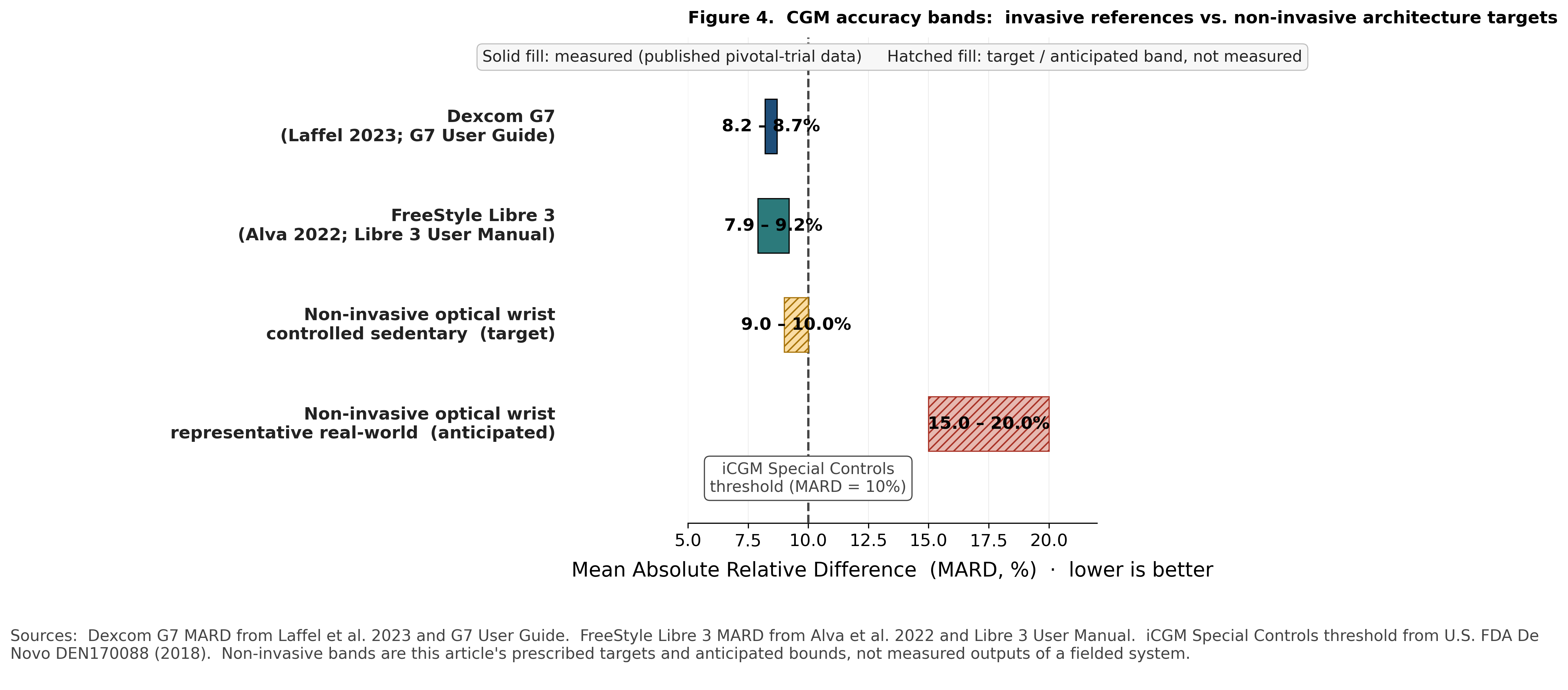
Figure 4. Accuracy bands across CGM categories: invasive references on the iCGM bar, non-invasive architecture targets dimensioned against and above it. Mean Absolute Relative Difference (MARD, %) for the four reference categories the article discusses. Solid-fill bars show measured Phase-3 trial outputs: Dexcom G7 at 8.2–8.7% MARD (Laffel et al. 2023, Diabetes Technol. Ther. 25:125; Dexcom G7 System User Guide) and Abbott FreeStyle Libre 3 at 7.9–9.2% MARD (Alva et al. 2022, Adv. Ther. 39:2839; Libre 3 User Manual). Hatched-fill bars show the article’s prescribed bands for the proposed non-invasive optical wrist architecture: a 9.0–10.0% target band in controlled sedentary cohorts, dimensioned against the FDA iCGM Special Controls accuracy threshold (10% MARD; FDA De Novo DEN170088, 2018; vertical dashed line), and a 15.0–20.0% anticipated band in representative real-world conditions accounting for the residual sensor-skin interface noise no architecture eliminates entirely. The gap between the controlled-sedentary target and the real-world anticipated band is owned, not denied; closing it across the deployment envelope is what Phase 3 engineering looks like for this device class. Hatched bars indicate target / anticipated values, not measured outputs of a fielded system.
No single breakthrough closes the gap on the non-invasive side. The architecture closes it by accumulating disciplined advantages across the stack.
Limitations and open problems
A credible architecture must also name what it cannot make disappear.
Showstopper: ISF–blood glucose lag. Interstitial and dermal glucose do not always track blood glucose instantaneously. During rapid transients after meals, insulin, or intense exercise, compartmental lag of 5 to 15 minutes matters. Existing CGMs already live with this. A non-invasive optical device will inherit the same constraint and must bound and communicate it clearly [9].
Showstopper: Photon budget at the long-wavelength edge. Water absorption climbs steeply beyond ~1700 nm. Beyond a certain depth, the shot-noise-limited SNR of even an idealized InGaAs detector drops below what the glucose signal needs. Source power cannot grow without bound; skin exposure and thermal safety impose limits. The architectural response is to confine quantitative wavelengths to the lower-absorption portion of the window and use the steeper-water region (1450 nm) only as a reference channel where SNR requirements are weaker.
Open problem: Long-term scattering stationarity. Skin scattering coefficients are characterized for single-time-point measurements. Whether at a fixed wrist site is stationary over a 7 to 10 day wear cycle, across hydration cycles, contact-induced micro-trauma, and circadian variation, is not established in the public literature. A non-stationary scattering coefficient redistributes pathlength under the diffusion model and shifts the effective extinction the constraint engine assumes. This is a longitudinal-data question the field has not yet answered.
Open problem: Universal vs. personalized calibration. Whether one calibration set learned across a representative population generalizes to a new subject’s skin type, body composition, and capillary geometry, or whether each subject requires a brief personalized calibration phase, is the second longitudinal question. The honest expectation, given the iCGM accuracy distributions, is that some form of personalized calibration window will be necessary, similar to the warm-up periods of current invasive CGMs. The architecture accommodates this. It does not eliminate it.
These are not reasons to dismiss the field. They are the boundary conditions for doing the field honestly.
Integration is the moat
Non-invasive glucose sensing is not an optics problem. It is not a silicon-photonics problem. It is not a physiology problem. It is not a packaging problem. It is not a machine-learning problem.
It is the simultaneous equation of all of them.
The optics team can choose a promising wavelength and still lose to skin variability. The physiology team can describe the tissue perfectly and fail to translate that knowledge into sensor geometry. The signal-processing team can build a beautiful model that succeeds in-sample and fails in the field. The packaging team can design a good contact interface and lose to photon budget. The AI team can build a powerful model and accidentally learn confounders.
Progress requires leaders and teams who can hold the whole stack in mind. Not because one person should do every discipline’s work. That is impossible. But someone must understand what each discipline is quietly assuming, what it is trading away, and how a choice upstream propagates five domains downstream.
That is the real moat. Not a secret wavelength. Not a proprietary algorithm. Not one clever mechanical design. The moat is the ability to integrate optics, tissue physiology, silicon photonics, thermal control, packaging, signal processing, machine learning, clinical validation, and manufacturing into one coherent architecture.
That is why the problem has remained open for so long.
And that is why it is finally becoming tractable.
The physics has not changed. The physiology has not changed. What has changed is the toolkit. Silicon photonics on CMOS, chip-scale narrow-linewidth sources, integrated detectors, low-power computation, better thermal sensing, better manufacturing, and physics-informed machine learning.
The generation that ships a truly non-invasive glucose monitor will not do it by wishing away the hard parts. It will ship by integrating them.
That is what makes the problem so difficult. It is also what makes it so interesting.
For an engineer who enjoys first-principles thinking, systems architecture, and the seams between disciplines, there may be no better problem in consumer sensing.
And if it is solved well, the impact will be enormous. Lower friction, lower consumable burden, broader metabolic awareness, and a path toward glucose monitoring that becomes accessible not only to diabetics but to anyone trying to understand their own physiology.
That is a problem worth solving.
About this article
This is a thought-leadership essay, not a peer-reviewed publication. Numerical claims (absorption coefficients, slopes, commercial-CGM MARDs, layer thicknesses) trace to the cited literature; architectural claims are the author’s first-principles reasoning, offered for critique rather than as established fact. The figures explicitly labeled “conceptual schematic” depict proposed design choices, not measured layouts. The author is responsible for any errors that have survived review. Material corrections will be noted in the revision history below with date and attribution to the reader who flagged them. Comments and corrections are welcome at ashutoshshastry.com or via LinkedIn. Views are the author’s own and do not represent the position of any employer.
Revision history
- v1.0 — April 26, 2026 — initial publication.
References
[1] K. Maruo et al., “In vivo noninvasive measurement of blood glucose by near-infrared diffuse-reflectance spectroscopy,” Appl. Spectrosc., vol. 57, no. 10, pp. 1236–1244, 2003.
[2] L. A. Coldren, S. W. Corzine, and M. L. Mashanovich, Diode Lasers and Photonic Integrated Circuits, 2nd ed. Wiley, 2012.
[3] J. Workman and L. Weyer, Practical Guide and Spectral Atlas for Interpretive Near-Infrared Spectroscopy, 2nd ed. CRC Press, 2012.
[4] M. A. Arnold, J. T. Olesberg, and G. W. Small, “Selectivity assessment of noninvasive glucose measurements based on analysis of multivariate calibration vectors,” J. Diabetes Sci. Technol., vol. 1, no. 4, pp. 454–462, 2007.
[5] T. J. Farrell, M. S. Patterson, and B. Wilson, “A diffusion theory model of spatially resolved, steady-state diffuse reflectance for the noninvasive determination of tissue optical properties in vivo,” Med. Phys., vol. 19, no. 4, pp. 879–888, 1992.
[6] U.S. Food and Drug Administration, “De Novo Classification Request for Dexcom G6 Continuous Glucose Monitoring System (iCGM), DEN170088,” 2018.
[7] L. M. B. Laffel et al., “Accuracy of a seventh-generation continuous glucose monitoring system in children and adolescents with type 1 diabetes,” Diabetes Technol. Ther., vol. 25, no. 2, pp. 125–132, 2023. (Plus Dexcom G7 System User Guide.)
[8] S. Alva et al., “Accuracy of the third generation of a 14-day continuous glucose monitoring system,” Advances in Therapy, vol. 39, pp. 2839–2852, 2022. (Plus Abbott FreeStyle Libre 3 System User Manual.)
[9] A. Heller and B. Feldman, “Electrochemical glucose sensors and their applications in diabetes management,” Chem. Rev., vol. 108, no. 7, pp. 2482–2505, 2008.
[10] T. L. Troy and S. N. Thennadil, “Optical properties of human skin in the near infrared wavelength range of 1000 to 2200 nm,” J. Biomed. Opt., vol. 6, no. 2, pp. 167–176, 2001.
[11] L. Kou, D. Labrie, and P. Chylek, “Refractive indices of water and ice in the 0.65–2.5 µm spectral range,” Appl. Opt., vol. 32, no. 19, pp. 3531–3540, 1993.
[12] D. M. Wieliczka, S. Weng, and M. R. Querry, “Wedge-shaped cell for highly absorbent liquids: infrared optical constants of water,” Appl. Opt., vol. 28, no. 9, pp. 1714–1719, 1989.
[13] M. Egawa, T. Hirao, and M. Takahashi, “In vivo estimation of stratum corneum thickness from water concentration profiles obtained with Raman spectroscopy,” Acta Derm. Venereol., vol. 87, pp. 4–8, 2007.
[14] J. Sandby-Møller, T. Poulsen, and H. C. Wulf, “Epidermal thickness at different body sites: relationship to age, gender, pigmentation, blood content, skin type and smoking habits,” Acta Derm. Venereol., vol. 83, pp. 410–413, 2003.
[15] S. L. Jacques, “Optical properties of biological tissues: a review,” Phys. Med. Biol., vol. 58, no. 11, pp. R37–R61, 2013.
Refs [1]–[9] are cited numerically in the article body. Refs [10]–[15] are cited in author-year form in the figure captions where the data they support is displayed; they are listed here for completeness.