Evanostics: silicon-photonics biosensor program from concept to clinical-stage
Tabletop silicon-photonics biosensor with four converging workstreams; the post-foundry surface-engineering chain later moved into a wearable at SiPhox
Evanostics, 2014–2021. A saliva-based diagnostic platform built across four parallel workstreams that converged at the interfaces. Eight granted US patents.

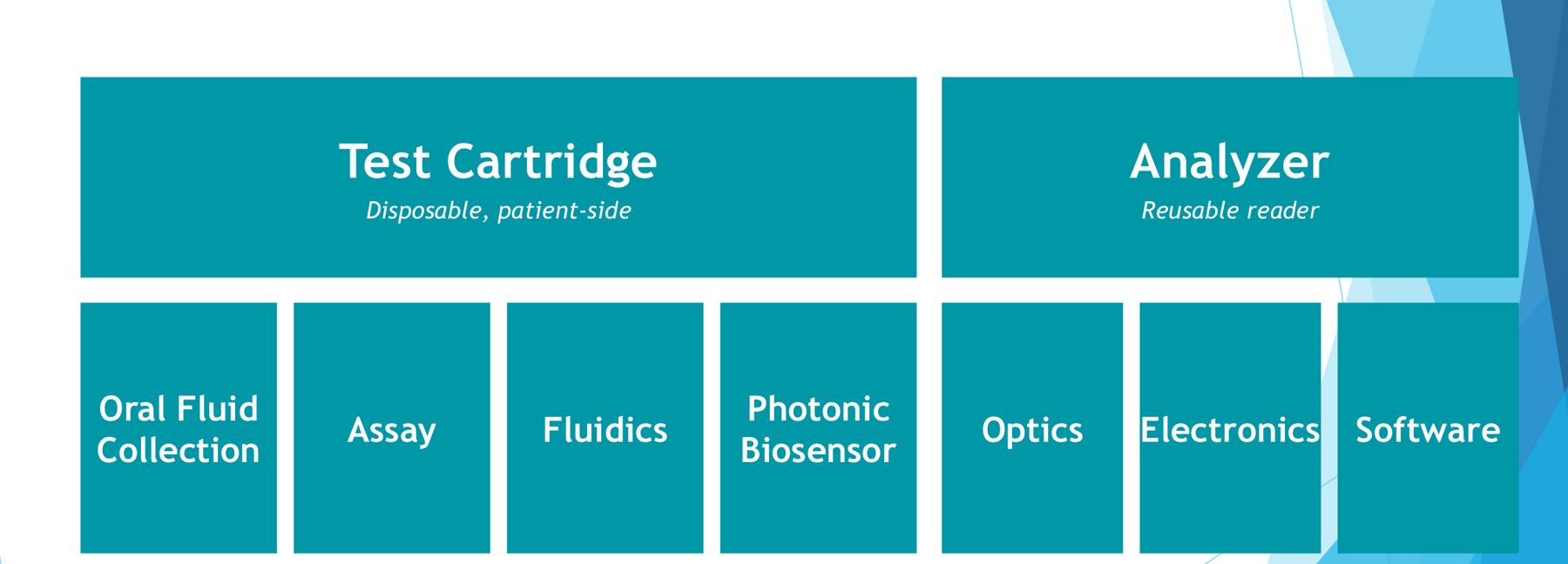
The product Evanostics built was a saliva-based oral-fluid diagnostic. The disposable cartridge was a single integrated consumable that combined the oral-fluid collection device, the sample-handling fluidics, the silicon-photonics chip, and the multiplex assay reagents, snapped into a clinical-grade reader. I joined as VP R&D, served as CTO across the program (the role under which the technical work below was led), and additionally as President and CEO for the final eight months.
The defining work
A platform built by four workstreams that had to converge: photonics and chip fabrication, biomolecule printing and a 10-plex competitive immunoassay (engineered with single-well normalization, on-chip positive controls, and signal amplification), modular fluidics (with the oral-fluid collection device as a substantial subsystem in its own right), and a clinical-grade reader. Each ran at its own cadence; integration happened at the interfaces. The work that defined the program was not any single technical breakthrough. It was building the common language and disciplined method by which a multi-disciplinary team named, resourced, and prioritized the interface problems where integration actually happens.
Outcome
Two IRB-governed clinical studies (2019, 2021); 335/342 concordance against LC-MS on a 38-patient cohort across nine drug classes; 13,000+ cartridges built through the pre-pilot chain; 95% yield on the final batch; pilot-line architecture designed for a step-up by an order of magnitude; eight granted US patents.
Sequel at SiPhox
The post-foundry process chain transferred. From 2021 to 2022, I led post-foundry process development at SiPhox Health for a silicon-photonics ring-resonator biosensor in a wearable form factor. The detection physics was different from Evanostics. Evanostics relied on evanescent-coupled fluorescence; SiPhox reads label-free shifts in ring-resonator frequency. What transferred was the post-fab surface-engineering chain itself: surface activation, silane functionalization, biomolecule deposition. The chemistry of Si-OH bond formation and silane attachment does not care which photonic architecture sits underneath.
Different detection physics, different form factor, same surface-engineering chain. Evanostics had been a tabletop reader where the core action lived on a 3-by-3 mm chip surrounded by empty space. SiPhox compressed silicon-photonics biosensing into a wearable. The harder problem is not the photonic core. It is everything around it. Fluidics, optics, electronics, packaging. What changes between form factors is how aggressively those layers integrate.